Copyright © 2012-20 Samaras & Associates, Inc., All Rights Reserved
Quality Management
OUR SERVICES: We can assist you with the preparation of your Design Control quality management system, we can train your personnel or audit your subcontractors, and we can help you manage compliance with the Design Control regulation. We also can assist you with the design, execution, analysis, and documentation of the required verifications and validation procedures, including software validations, for your specific type of medical device. Additionally, we can provide you with the complete set of technical management consulting services necessary to better integrate your engineering functions with the rest of your business, improving productivity and performance. OUR PRINCIPALS: One of our principals is an ASQ-certified Quality Engineer and Biomedical Auditor with a doctorate in Engineering Management and is a former FDA reviewer & manager with extensive experience drafting, developing, and helping clients manage, medical device quality management systems. The design, development, manufacturing, and servicing of medical devices
marketed in the US are regulated by the FDA's Quality System regulation (21
CFR 820). This regulation applies to all medical devices of all classes, as
appropriate (e.g., some Class 1 devices only have to comply with certain parts
of the regulation). One subpart of the regulation describes Design Controls (21
CFR 820.30), which focuses on the medical device development process.
Compliance with Design Controls is required of all Class 2 and Class 3 medical
devices AND any devices automated with computer software. You will have to
certify compliance in your 510(K) submission (21 CFR 807.87(k)); you will be
audited by the FDA District Office for your PMA submission.
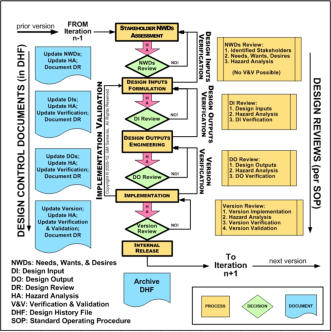
Design Controls are derived from the principles of classical systems
engineering. They define the medical device development lifecycle required by
FDA regulation (see recent article by one of our principals). The upper figure
on the right shows this in schematic form; a simpler view may be found here.
The Quality System regulation not only requires compliance with Design
Controls, but also requires a documented quality management system
(consisting of Standard Operating Procedures, Work Instructions, and Forms &
Templates) that specify how Design Controls will be managed (i.e.,
implemented, measured, and controlled) in your organization.
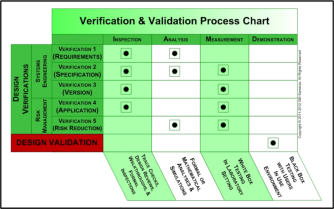
Two elements of Design Controls create confusion and difficulty; these are
engineering validation and engineering verifications. In the medical device
design control schema, validation means that you developed the right system
(the developers correctly solved the problem captured by the design input
process); verification means that you developed the system the right way (the
developers did their job correctly at each step of the process). The middle
figure on the right shows that, while there can only be one type of design
validation, there are five (5) different types of design verifications.
Three types of engineering design verification are from classical systems
engineering (verification of design inputs, design outputs, and design
implementation). The other two types of verification are from risk
management; they are verification that you properly applied your proposed
mitigation(s) and verification that your properly applied mitigation actually
reduced the target risk(s). Unlike validation, verifications are internal error-
correcting mechanisms in the engineering process that reduce the probability
of mistakes during the development, deployment, or maintenance effort.
Validation lets you determine whether you developed what your stakeholders
asked you to develop (in the design inputs) - did you develop the right system?
Validation can occur at the system level, the subsystem level (e.g., software
validation, usability validation), or the module level (e.g., validation of 3rd party
modules designed for interoperability). System validation is your final defense
against Type III errors: "correctly solving the wrong problem".
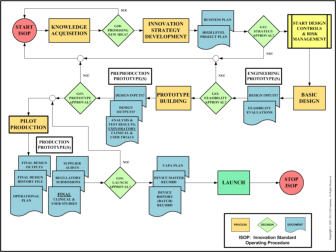
Validation and the 5 verifications are all about avoiding mistakes in the
engineering development of your medical device. They are a critical part of the
medical device development schema (see lower figure on right), which is an
important consideration when operating in a regulated industry and central to
effective technical management.
All testing needs to be statistically sound.
The design, development, manufacturing, and servicing of medical devices
marketed in the US are regulated by the FDA's Quality System regulation (21
CFR 820). This regulation applies to all medical devices of all classes, as
appropriate (e.g., some Class 1 devices only have to comply with certain parts
of the regulation). One subpart of the regulation describes Design Controls (21
CFR 820.30), which focuses on the medical device development process.
Compliance with Design Controls is required of all Class 2 and Class 3 medical
devices AND any devices automated with computer software. You will have to
certify compliance in your 510(K) submission (21 CFR 807.87(k)); you will be
audited by the FDA District Office for your PMA submission.
Design Controls are derived from the principles of classical systems
engineering. They define the medical device development lifecycle required by
FDA regulation (see recent article by one of our principals). The upper figure
on the right shows this in schematic form; a simpler view may be found here.
The Quality System regulation not only requires compliance with Design
Controls, but also requires a documented quality management system
(consisting of Standard Operating Procedures, Work Instructions, and Forms &
Templates) that specify how Design Controls will be managed (i.e.,
implemented, measured, and controlled) in your organization.
Two elements of Design Controls create confusion and difficulty; these are
engineering validation and engineering verifications. In the medical device
design control schema, validation means that you developed the right system
(the developers correctly solved the problem captured by the design input
process); verification means that you developed the system the right way (the
developers did their job correctly at each step of the process). The middle
figure on the right shows that, while there can only be one type of design
validation, there are five (5) different types of design verifications.
Three types of engineering design verification are from classical systems
engineering (verification of design inputs, design outputs, and design
implementation). The other two types of verification are from risk
management; they are verification that you properly applied your proposed
mitigation(s) and verification that your properly applied mitigation actually
reduced the target risk(s). Unlike validation, verifications are internal error-
correcting mechanisms in the engineering process that reduce the probability
of mistakes during the development, deployment, or maintenance effort.
Validation lets you determine whether you developed what your stakeholders
asked you to develop (in the design inputs) - did you develop the right system?
Validation can occur at the system level, the subsystem level (e.g., software
validation, usability validation), or the module level (e.g., validation of 3rd party
modules designed for interoperability). System validation is your final defense
against Type III errors: "correctly solving the wrong problem".
Validation and the 5 verifications are all about avoiding mistakes in the
engineering development of your medical device. They are a critical part of the
medical device development schema (see lower figure on right), which is an
important consideration when operating in a regulated industry and central to
effective technical management.
All testing needs to be statistically sound.